Shortening the path to rare disease diagnosis by using newborn genetic screening and digital technologies
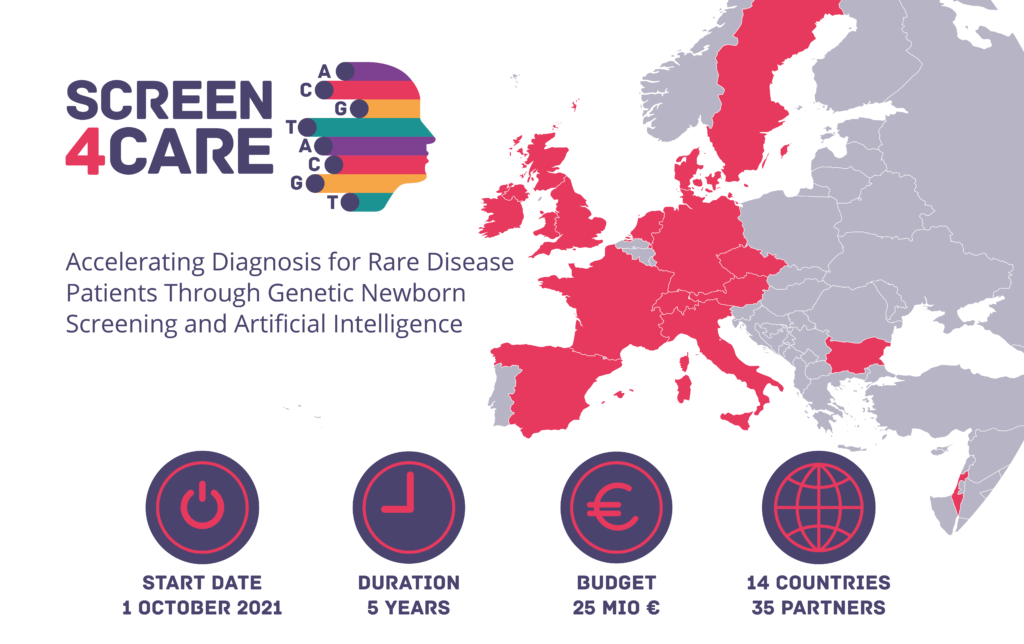
New EU Research Project “Screen4Care”: Accelerating Diagnosis for Rare Disease Patients Through Genetic Newborn Screening and Artificial Intelligence
The international consortium aims to tackle the major hurdle for rare disease patients – the lengthy and convoluted diagnosis journey – via an innovative research approach based on two central pillars: genetic newborn screening and artificial intelligence (AI)-based tools.
On 1 October 2021 an international public-private consortium of 35 partners announced the launch of Screen4Care – a research project that aims to significantly shorten the time required for rare disease diagnosis and efficient intervention by utilising genetic newborn screening and advanced analysis methods such as machine learning. The project will run for a period of five years with a total budget of EUR 25 million provided by the Innovative Medicines Initiative (IMI 2 JU), a joint undertaking of the European Union and the European Federation of Pharmaceutical Industries and Associations (EFPIA).
Motivation
There are more than 7,000 known rare diseases, conditions that affect one, or less than one, person in 2,000. These conditions, which collectively impact an estimated 27-36 million people across the EU and will affect one in 17 people during their lifetime, are often severe, multisystemic chronic diseases that put patients at risk of permanent organ damage and degeneration. Patients typically face an arduous journey to proper diagnosis, enduring on average eight years of countless doctor’s consultations, misdiagnoses and ineffective treatments. Lengthy diagnosis journeys place a heavy burden on patients, their families and society. They also hinder swift intervention – such as appropriate treatments or enrolment in clinical trials – and patient empowerment, realised through strategies such as lifestyle adjustments, family planning, genetic counselling and coping with the psychosocial and/or financial consequences of the condition.
Screen4Care will use a multi-pronged strategy to shorten the time to diagnosis and treatment for patients with rare diseases:
- Genetic newborn screening: The project will drive newborn screening (using genetic testing and related advanced genomic technologies), which is anticipated to be an effective tool for early diagnosis given that approximately 72% of rare diseases have a genetic cause and 70% of rare disease patients are children.
- AI-based tools: The project will design and develop new AI algorithms to identify patients at early disease onset via electronic health records and develop a repository of AI ‘symptom checkers’ to help patients who are in the midst of their diagnosis journeys – both supporting symptom-based diagnosis later in life.
Project goals
In addition to its goal of developing the core early-diagnosis system, Screen4Care aims to establish a digital infrastructure and ecosystem to engage patients, parents of newborns and caregivers as equal decision-makers in the diagnosis process. The ecosystem will provide an open innovation platform, which allows for continuous data collection and information exchange, aiding the development of next-generation diagnostics and enabling physicians, patients and relatives to make informed decisions at an earlier stage. Screen4Care proposes that this will contribute to minimised disease progression, improved patient health and quality of life and an optimised use of healthcare resources.
Project Partners
Project Lead
Pfizer Ltd, UK
Austria
- Ludwig Boltzmann Gesellschaft GmbH
- Research Institute AG & Co KG
- SBA Research gemeinnützige GmbH
Bulgaria
- Bulgarian Association for Personalized Medicine
- Bulgarian Association for the Promotion of Education and Science
Czech Republic
- University Karlova
Denmark
- Copenhagen Business School
- FindZebra ApS
- Novo Nordisk A/S
- Syddansk University
France
- Eurordis-Rare Diseases Europe
- Lysogene S.A.
- Sanofi-Genzyme
Germany
- Charité – Universitätsmedizin Berlin
- Eurice – European Research and Project Office GmbH
- Max-Planck-Gesellschaft zur Förderung der Wissenschaften e.V.
- Universitätsmedizin Göttingen – Georg-August-Universität Göttingen
- University Hospital Bonn
- University Hospital Erlangen
Ireland
- University College Dublin, National University of Ireland (Inclusive Design Research Centre-SMARTlab)
Israel
- Genoox
Italy
- Consorzio Futuro in Ricerca
- Pediatric Hospital Bambino Gesù
- University of Ferrara
- University of Siena
Spain
- Centro Nacional de Análisis Genómico (CNAG-CRG)
Sweden
- Uppsala University
Switzerland
- F. Hoffmann-La Roche AG
- sitem-insel AG
- Novartis Pharma AG
- Takeda Pharmaceuticals International AG
- University Bern
The Netherlands
- ProQR Therapeutics NV
United Kingdom
- Illumina Cambridge Ltd
- Pfizer Ltd
Further information
- More info could be found here: https://www.screen4care.eu
Contact
The project will run for a period of five years with a total budget of EUR 25 million provided by the Innovative Medicines Initiative (IMI 2 JU), a joint undertaking of the European Union and the European Federation of Pharmaceutical Industries and Associations (EFPIA).